medical devices
biocompatible solutions for optimized performance
When it comes to medical devices biocompatibility, compliance, and patient safety are the key requirements. Whether it is a hemocompatible surface technology, a customizable adhesive and potting compound, or a phthalate-free plasticizer, Aurorium helps medical device customers optimize device performance and improve manufacturing efficiency. Backed by regulatory and application experts, Aurorium solutions are approved by global regulatory bodies and are used by leading medical device manufacturers worldwide.

A clinically proven, time-tested, truly multifunctional surface technology, PC Technology™ imparts to the device surface antithrombotic, anti-inflammatory, hydrophilic, and antimicrobial properties that reduce biofilm formation and subsequent fouling and encrustation.
learn more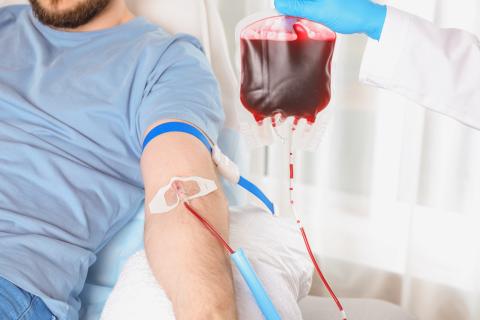
Citroflex™ B-6 phthalate-free plasticizer offers excellent performance in medical PVC plastics formulations. With a clean toxicological profile, Citroflex B-6 presents a proven, field-tested alternative to traditional DEHP plasticizers.
learn more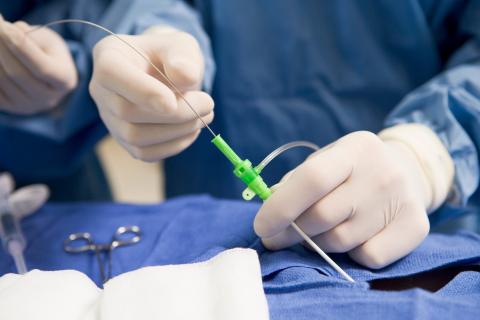
Biothane™ Polyurethane Systems can be custom formulated to provide the optimum pot life, cure time, strength, and flexibility required to fit the unique needs of catheters and specialty medical device assembly processes.
learn more
DCDPS (4,4′-Dichlorodiphenyl Sulfone) is a monomer used for polymerization into the production of polysulfones (PES). High-performance sulfones are categorized by high transparency and high-temperature performance. Sulfones are used as replacements for metals and glass in medical products.